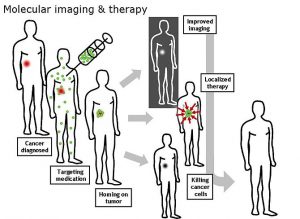
Cancer cells are incredibly flexible about promoting their own movement and growth in the body. They can travel through blood vessels as thin as spider silk. They even change their shape to do so, yet are still able to divide and cluster into colonies in those very skinny spaces. That spreading through the body is called metastasis, and it’s what makes cancer turn deadly.
Researchers are now putting nanotechnology to work to help decipher exactly how cancer cells perform this extraordinary feat. An article in Nanotechnology Now reports:
The researchers trapped live cancer cells in the tubular membranes and, with optical high- and super-resolution microscopy, could see how the cells adapted to the confined environment. Cell structures significantly changed in the nanomembranes, but it appeared that membrane blebbing — the formation of bulges — at the cells’ tips helped keep genetic material stable, an important requirement for healthy cell division.
For more details, check out this one-minute video on how scientists used microtubular membranes to study how cancer cells divide in capillaries.
Related articles